Toegevoegd aan je winkelmand!

IKA BMT-20-S-IVD.50 Ball milling tube, sterile, IVD label
€883,00
Toegevoegd aan je winkelmand!

IKA BMT-20-S-IVD1.50 Ball milling tube, sterile, IVD label
€690,00
Toegevoegd aan je winkelmand!

IKA UTTD control Pharma Solution
€1.726,00
Toegevoegd aan je winkelmand!

IKA UTTD control Life Science Solution
€1.743,00
Toegevoegd aan je winkelmand!
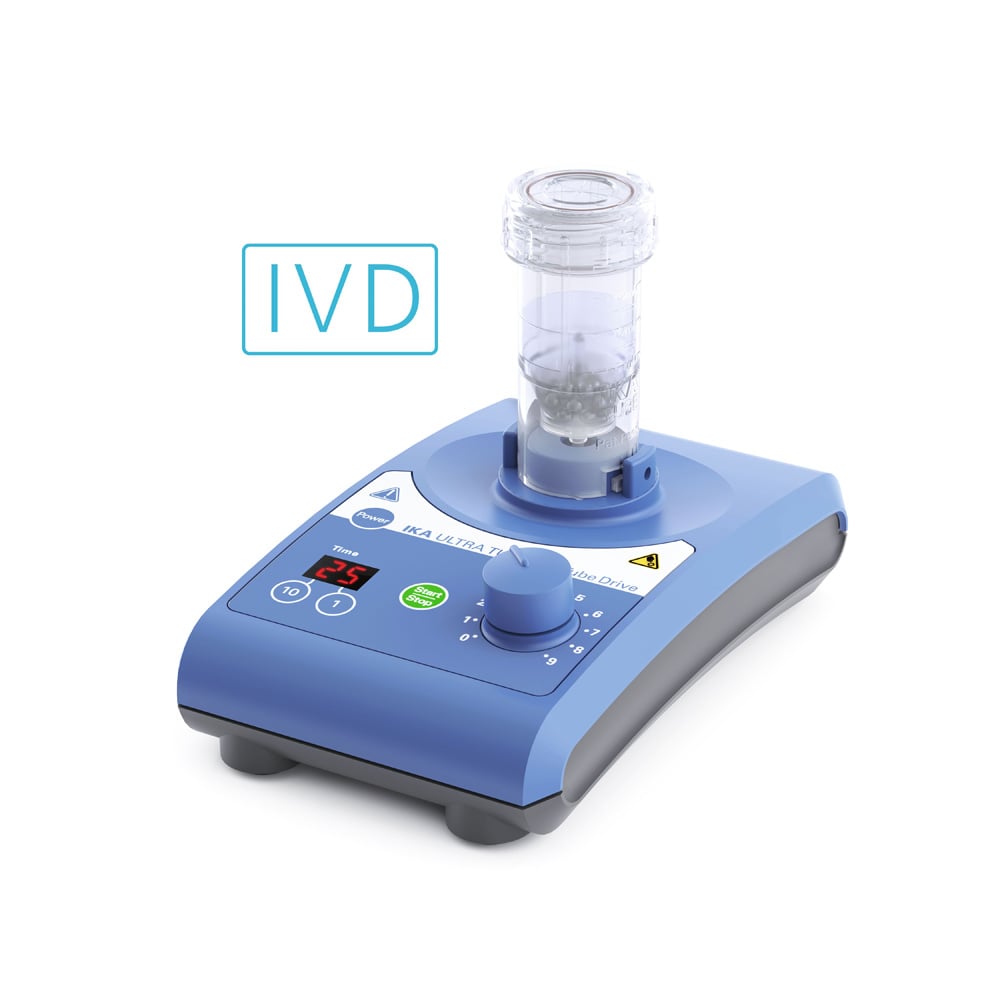
IKA UTTD basic IVD Solution Maalmolen
€1.229,00
Microbiological and molecular biological analysis of patient samples. The crushing and breaking down of tissue, cartilage and bone samples is an essential step before the actual microbiological and molecular biological analysis of patient samples.
The challenges in this pre-operative step are to crush the sample homogeneously and not to contaminate the sample on the way to analysis.
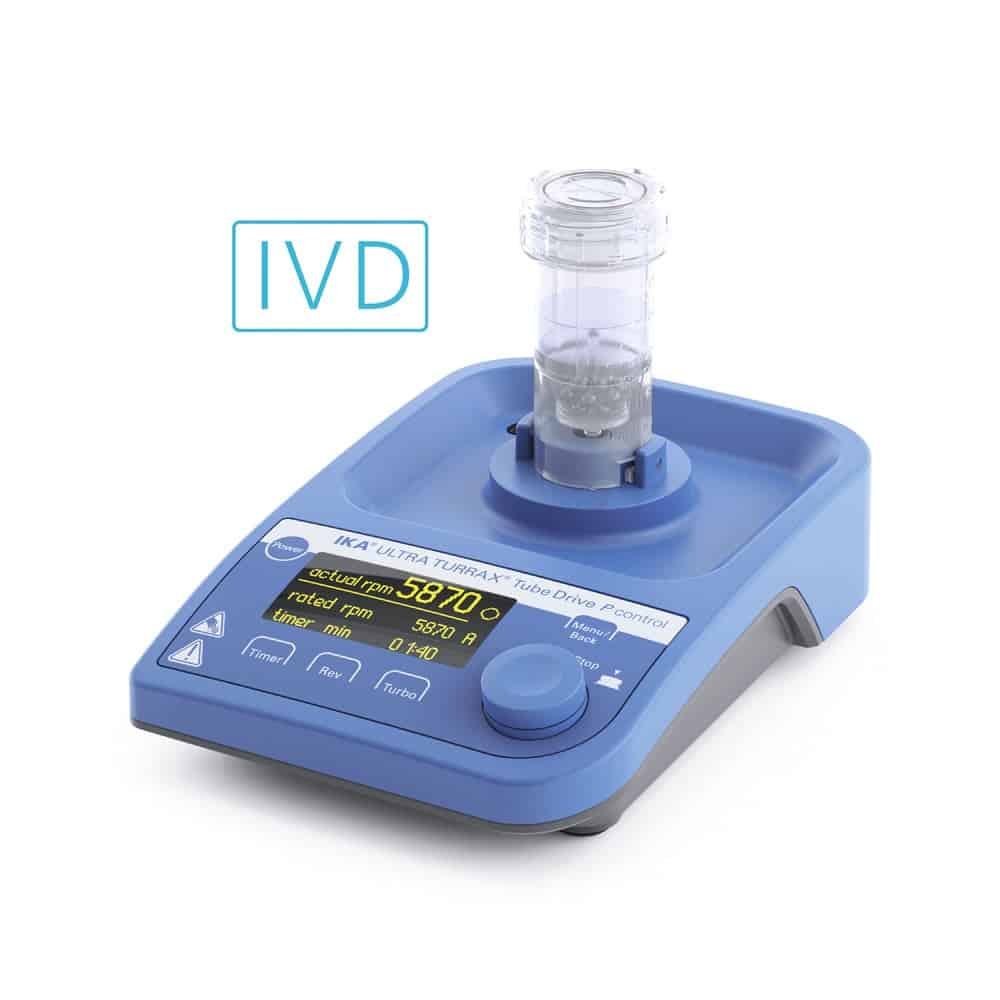
With the ULTRA-TURRAX® Tube Drive P control, IKA offers a powerful universal dispersing drive that is operated in combination with patented disposable plastic tubes.
The sample tubes specially developed for in-vitro diagnostics are available in single or double sterile packaging and are already filled with 5 ml of 0.90% saline solution.
The sample is crushed with 10 stainless steel balls contained in the tube, which are set in motion with the help of the drive and a tumbler with up to 3000 revolutions. The accelerated balls crush the sample or reliably detach tissue from the bone. Any germs that may be present pass into the liquid phase (NaCl solution).
The sample material can be introduced into the sterile sample container directly in the operating room by opening the screw cap from the lid. The transport in the closed tube is hermetically sealed without the risk of cross-contamination. In the microbiological laboratory, the tube is placed directly on the drive and the sample is crushed. The samples can be taken via the pierceable membrane in the sample lid.
The lockable disposable sample tubes are transport containers, crushing containers and storage all in one. They reduce the cleaning effort and systematically rule out cross-contamination. Users benefit from reduced handling costs and a reduced number of incorrect diagnoses.
In the control variant of the ULTRA-TURRAX® Tube Drive, process parameters such as time and speed can be stored as a program - this ensures that samples are processed consistently. The direction of rotation can be reversed for demanding samples. Tests can be documented via a PC and the appropriate software and started automatically.
The disposable tubes BMT-20-S-IVD included in the package are a Invitro diagnostic product according to Directive 98/79/EG and manufactured according to ISO 13485:2016. Approved for all EU countries.
Important note: The maximum processing time for the tubes is 1 minute using an automated process similar to the ULTRA-TURRAX® Tube Drive. If the processing time is longer, the mechanical load can become too great and leaks can occur. The process must be validated by the customer.
Please also note the operating instructions for the ULTRA-TURRAX® Tube Drive.
Scope of delivery: ULTRA-TURRAX® Tube Drive P control | BMT-20-S-IVD.50 Ball milling tube, sterile, IVD label, 20 ml
| Motor rating input [W] | 36 |
| Motor rating output [W] | 28 |
| Speed display | OLED |
| Speed range [rpm] | 400 - 8000 |
| Viscosity max. [mPas] | 5000 |
| Timer | yes |
| Timer display | OLED |
| Speed deviation [%] | 3 |
| Speed adjustment | 10 RPM Steps |
| Noise without element [dB(A)] | 50 |
| Process type | batch |
| Reversible direction of rotation | yes |
| Dimensions (W x H x D) [mm] | 122 x 54 x 178 |
| Weight [kg] | 1.3 |
| Permissible ambient temperature [°C] | 5 - 40 |
| Permissible relative humidity [%] | 80 |
| Protection class according to DIN EN 60529 | IP 20 |
| USB interface | yes |
| Voltage [V] | 100 - 240 |
| Frequency [Hz] | 50/60 |
| Power input [W] | 36 |
| DC Voltage [V=] | 24 |
| Current consumption [mA] | 1500 |
Microbiological and molecular biological analysis of patient samples. The crushing and breaking down of tissue, cartilage and bone samples is an essential step before the actual microbiological and molecular biological analysis of patient samples.
The challenges in this pre-operative step are to crush the sample homogeneously and not to contaminate the sample on the way to analysis.
With the ULTRA-TURRAX® Tube Drive P control, IKA offers a powerful universal dispersing drive that is operated in combination with patented disposable plastic tubes.
The sample tubes specially developed for in-vitro diagnostics are available in single or double sterile packaging and are already filled with 5 ml of 0.90% saline solution.
The sample is crushed with 10 stainless steel balls contained in the tube, which are set in motion with the help of the drive and a tumbler with up to 3000 revolutions. The accelerated balls crush the sample or reliably detach tissue from the bone. Any germs that may be present pass into the liquid phase (NaCl solution).
The sample material can be introduced into the sterile sample container directly in the operating room by opening the screw cap from the lid. The transport in the closed tube is hermetically sealed without the risk of cross-contamination. In the microbiological laboratory, the tube is placed directly on the drive and the sample is crushed. The samples can be taken via the pierceable membrane in the sample lid.
The lockable disposable sample tubes are transport containers, crushing containers and storage all in one. They reduce the cleaning effort and systematically rule out cross-contamination. Users benefit from reduced handling costs and a reduced number of incorrect diagnoses.
In the control variant of the ULTRA-TURRAX® Tube Drive, process parameters such as time and speed can be stored as a program - this ensures that samples are processed consistently. The direction of rotation can be reversed for demanding samples. Tests can be documented via a PC and the appropriate software and started automatically.
The disposable tubes BMT-20-S-IVD included in the package are a Invitro diagnostic product according to Directive 98/79/EG and manufactured according to ISO 13485:2016. Approved for all EU countries.
Important note: The maximum processing time for the tubes is 1 minute using an automated process similar to the ULTRA-TURRAX® Tube Drive. If the processing time is longer, the mechanical load can become too great and leaks can occur. The process must be validated by the customer.
Please also note the operating instructions for the ULTRA-TURRAX® Tube Drive.
Scope of delivery: ULTRA-TURRAX® Tube Drive P control | BMT-20-S-IVD.50 Ball milling tube, sterile, IVD label, 20 ml
Toebehoren
Serie
FAQ
Heb je vragen over dit product of over deze categorie? Op onze FAQ-sectie geven we je antwoord op veelgestelde vragen!
Bekijk de FAQJe kan onze experts altijd bereiken via info@imlab.eu
Of bel ons direct op +32 (0) 16 73 55 72
(Bereikbaar op werkdagen van 08:00 tot 17:00)

Stel direct je vraag
"*" geeft vereiste velden aan
Onze diensten – méér dan een webshop
Bij Imlab koop je niet zomaar een product – je kiest voor een betrouwbare partner met een team dat voor je klaarstaat.
Geen robots, geen callcenter, maar ervaren specialisten die je helpen.
Onze service omvat:
- Kalibratie van je meetapparatuur
- Onsite installatie
- Herstellingen & onderhoud
Hulp nodig of een service aanvragen? We denken graag met je mee en bieden oplossingen op maat, zodat jij snel verder kan.